














| Customization: | Available |
|---|---|
| Application: | Biotechnology Industry, Chemical industry, Clinical Diagnosis, Food & Beverage Factory, Food Safety, Forensic Science, Pharmaceutical Industry |
| Capacity: | 20000 L/hr |
Solution Preparation System CIP/SIP Integrated Mixing Equipment with Mixing Tank
The Solution Preparation Mixing Equipment is a specialized pharmaceutical apparatus designed for liquid mixing and dispensing operations. This equipment enables the preparation of pharmaceutical solutions by combining active pharmaceutical ingredients with solvents such as water for injection. Through integrated processes including controlled stirring, heating, sterilization, and filtration, the system produces medicinal solutions at precise target concentrations. These systems find application across both sterile and non-sterile pharmaceutical manufacturing environments.


Classification
✅ 1) Modular Design - Cost-Saving & Efficient
Pre-tested modular equipment reduces installation costs, accelerates delivery, and ensures flexibility.

✅ 2) Fully Automated Operation - Smart & Compliant
1-click CIP/SIP with auto-detection for cleaning/sterilization completion. Computerized features: Recipe management, e-signature, e-records, and audit trail for full automation.
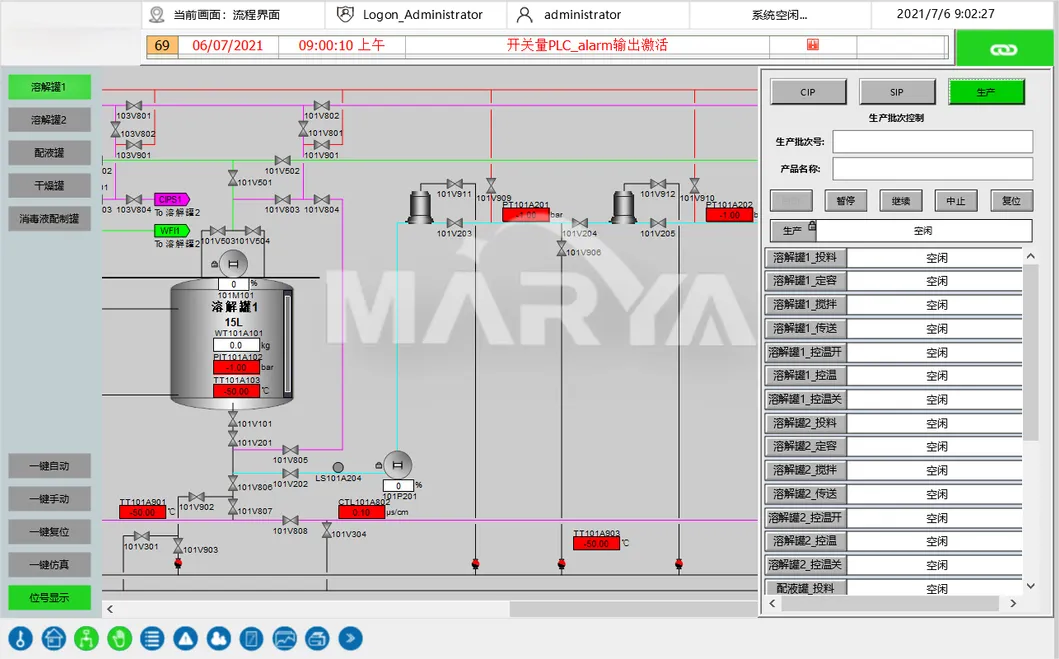
✅ 3) Process Control & Real-Time Monitoring
Critical parameter control (temp, DO, pH) with auto-alerts for deviations to ensure drug quality.

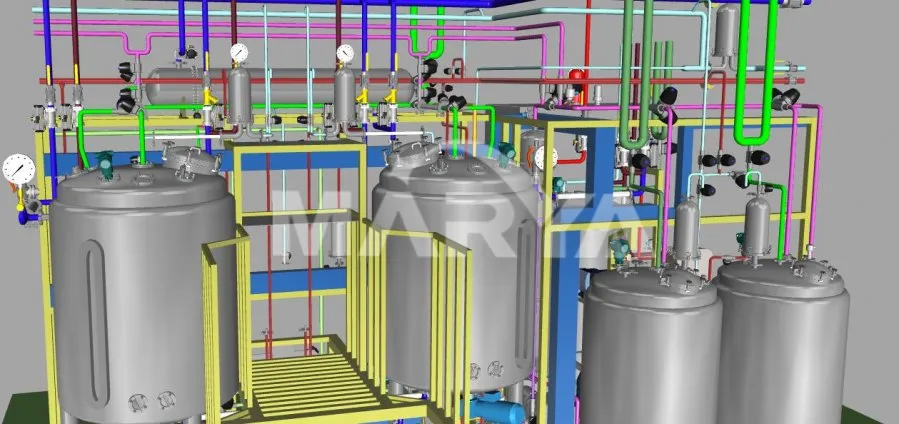
✅ 4) 3D Simulation & Ergonomic Layout
Pre-production 3D modeling optimizes piping, valve, and instrument placement for user-friendly operation.
| Name | Parameters |
|---|---|
| Working volume | 2~15000L |
| Material | 304, 316L, 904L, TA2 |
| Stirring type | Upper mechanical, lower magnetic stirring |
| Stirring speed control | 0~450rpm ±3.0% |
| Temperature control | -10~150ºC ±0.2ºC |
| Pressure control | -0.01Mpa~0.06Mpa ±0.01MPa |
| Weighing control | 3‰ |
| Protection level | IP65 |










 Coden Kitchen
Coden Kitchen